|
The false-negative rRT-PCR test probability for SARS-CoV-2 depends on various sampling and technical factors, whereas the chances of obtaining a true positive result decreases over time, and with decreasing viral titers in clinical specimens. Retrospectively, test positivity does not always show an infection existing in reality. One or more negative tests do not rule out the possibility of SARS-CoV-2 infection. It is widely accepted that tests are not completely foolproof, and, thus no single ‘gold standard’ assay exists. Nowadays, immunometric assays (IMAs) are performed for detecting the immune response to COVID-19, while several test manufacturers have launched various rapid diagnostic tests (RDTs), to facilitate SARS-CoV-2 detection at point-of-care. As defined by World Health Organization (WHO), a confirmed case is detected from nucleic acid amplification tests (NAAT) for SARS-CoV-2, such as real-time reverse transcription polymerase chain reaction (rRT-PCR), that is worldwide preferred. 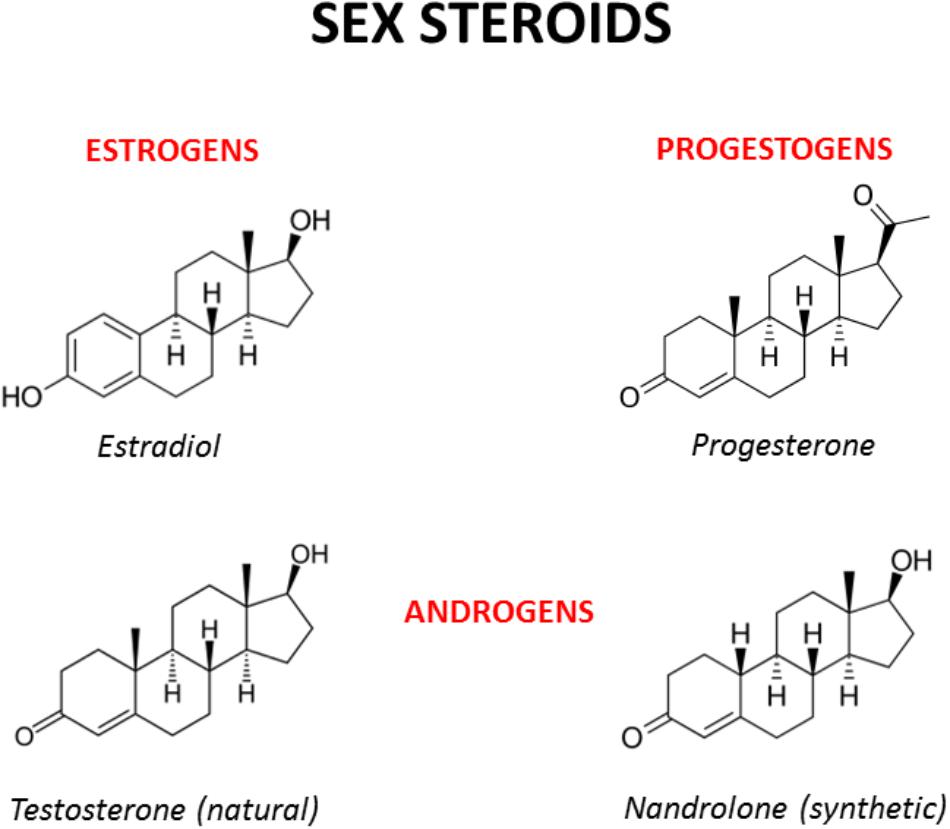
In December 2019, a novel coronavirus (SARS-CoV-2) was reported from a cluster of pneumonia cases in Wuhan, China. Three highly pathogenic coronaviruses have impacted substantially on human populations since the beginning of the 21st century. Future avenues of research to evaluate lung tissue innate immune responses hold promise as a target for research to optimize SARS-CoV-2 and future infections’ testing accuracy. Prior immunization against SARS-CoV-2 impairs the utility of serologic testing of suspected COVID-19 cases. Automated testing platforms may enhance diagnostic accuracy by minimizing the potential for human error in assays’ performance. Different COVID-19 management algorithms based on each test and limitations are thoroughly presented.Įxpert opinion: The diagnostic accuracy and the capacity of every available assay, which need to be interpreted in the light of the background incidence of SARS-CoV-2 infection in the communities in which they are used, are essential in order to minimize the number of falsely tested cases. How clinical characteristics, specific respiratory comorbidities and SARS-CoV-2 vaccination impact on existing diagnostic assays are highlighted. 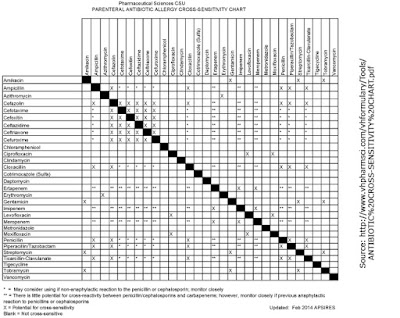
Accuracy of SARS-CoV-2 diagnostic assays has emerged as a major barrier to COVID-19 diagnosis, particularly in cases requiring urgent or emergent treatment.Īreas covered: In this review, we explore the major reasons for false-positive and false-negative SARS-CoV-2 test results. Introduction: A novel Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) was reported via nucleic acid identification in December, 2019.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
